|
12/14/2023 0 Comments Palmetto retina center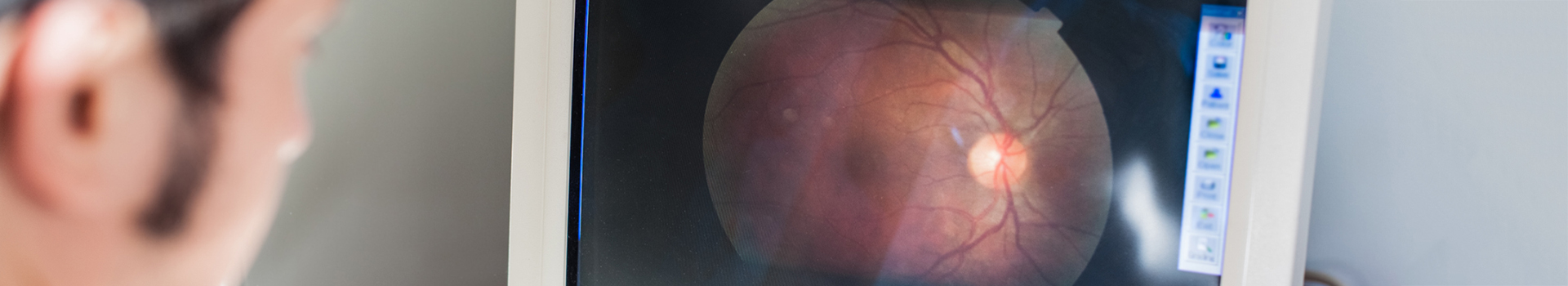 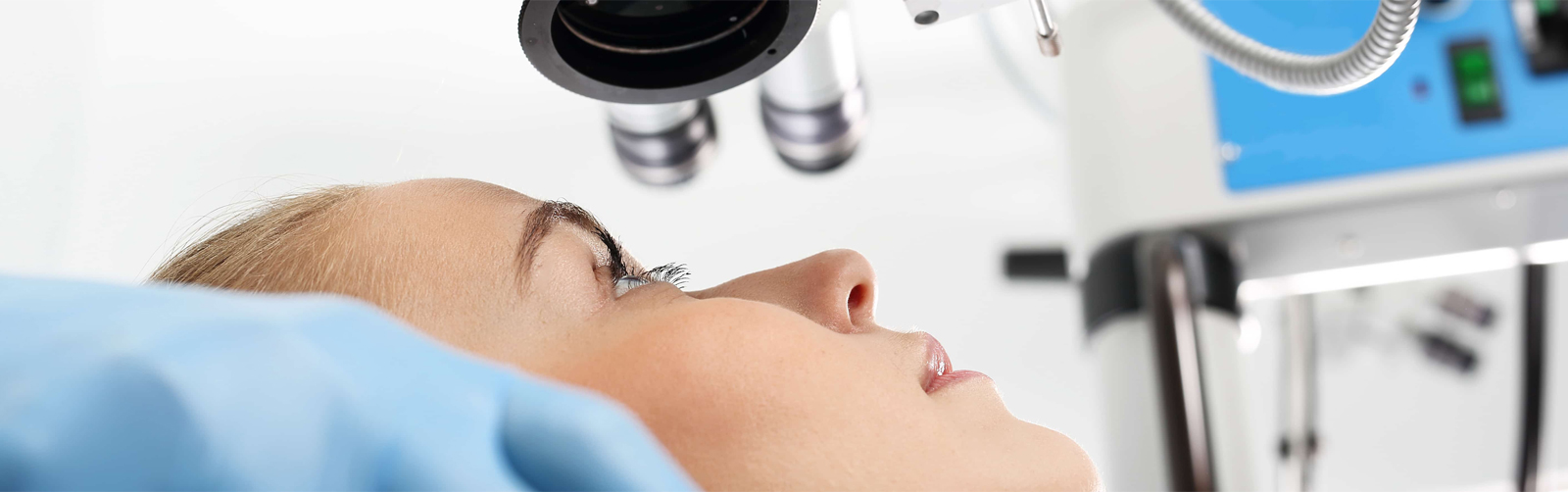
“Having a new therapy for which approximately half of patients were able to be safely treated as infrequently as every 4 months is a big step forward for patients with chronic macular diseases like nAMD and DME-common conditions that traditionally require very frequent injections (every 1 to 2 months) with standard anti-VEGF monotherapy alone,” he said. Moshfeghi noted that faricimab appears to offer patients the chance to have more robust control of their macular disease and the potential for a longer treatment benefit. Moshfeghi, MD, MBA, associate professor of ophthalmology, University of Southern California. “The forthcoming availability of faricimab is a refreshing way to kick off 2022 for retina specialists and patients with macular disease alike,” said Andrew A. “This drug should ease the treatment burden on our patients with Wet AMD and DME.” “After giving presentations of data from the studies, and witnessing those presented by other investigators It is clear to me that this drug offers impressive efficacy and extended duration of action combined with an excellent safety profile,” he said. Lawrence Singerman, MD, FACS, of Retina Associates of Cleveland and a clinical professor of ophthalmology at Case Western Reserve University and Bascom Palmer Eye Institute, said he has been anticipating the announcement of FDA approval for faricimab. “This is our second FDA approval in ophthalmology in recent months, underscoring our commitment to people living with retinal conditions,” he added in the statement.įor retina specialists, the approval is welcome news and offers a new treatment option for patients. In the release, Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, faricimab “provides a new approach to treating vision-threatening retinal conditions through a mechanism of action that targets two pathways simultaneously.” 
1 Additionally, the FDA is reviewing Genentech’s application fordiabetic retinopathy. Genentech also submitted a Marketing Authorization Application for faricimab for the treatment of nAMD and DME, which the European Medicines Agency has accepted. “With Vabysmo, we now have the opportunity to offer patients a medicine that could improve their vision, potentially lowering treatment burden with fewer injections over time.” It is the first bispecific antibody approved for the eye and a major advance in treating retinal conditions such as wet AMD and diabetic macular edema,” Charles Wykoff, MD, PhD, director of research at Retina Consultants of Texas in Houston and a faricimab Phase III investigator, said in a statement. “Vabysmo represents an important step forward for ophthalmology. Faricimab is the first and only FDA-approved drug to target two distinct pathways, Ang-2 and VEGF-A, that often cause retinal disease that may lead to visual loss. 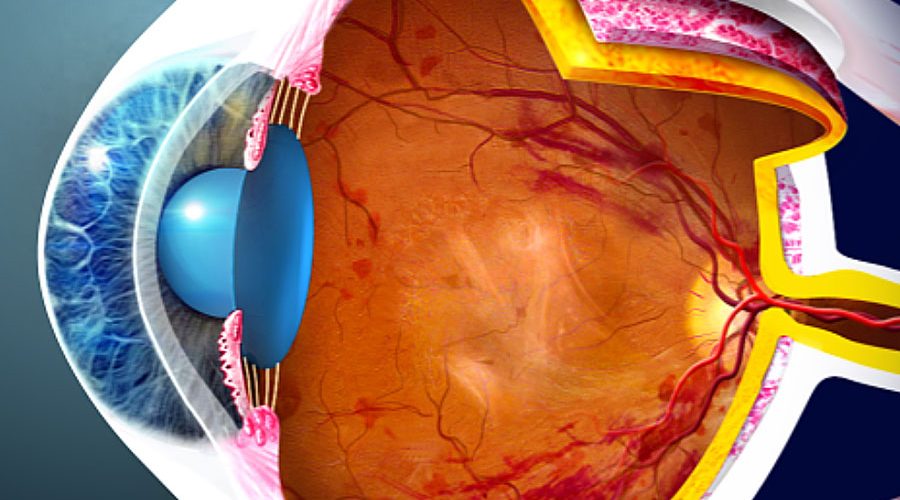
Food and Drug Administration (FDA) approved Vabysmo™ (faricimab-svoa, Genentech), a bispecific antibody for the treatment of wet age-related macular degeneration (AMD) and diabetic macular edema (DME).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
